conductor
What is a conductor?
A conductor, or electrical conductor, is a substance or material that allows electricity to flow through it. In a conductor, electrical charge carriers, usually electrons or ions, move easily from atom to atom when voltage is applied. Most metals like copper are considered good conductors, while nonmetals are considered bad conductors -- that is, insulators.
Understanding electrical conductors
In general, conductivity refers to the capacity of a substance to transmit electricity or heat. A conductor conducts electricity since it offers little or no resistance to the flow of electrons, thus leading to a flow of electrical current. Typically, metals, metal alloys, electrolytes and even some nonmetals, like graphite and liquids, including water, are good electrical conductors. Pure elemental silver is one of the best electrical conductors. Other good electrical conductors include the following:
- copper
- steel
- gold
- silver
- platinum
- aluminum
- brass
Human beings are also good conductors of electricity, which is why touching someone experiencing an electric shock causes the toucher to experience the same shock. In electrical and electronic systems, conductors comprise solid metals molded into wires or etched onto printed circuit boards.
Key characteristics of electrical conductors
Important features of an electrical conductor include the following:
- It ensures free movement of electrons or ions through it.
- It has a zero electric field inside, which permits the movement of electrons or ions.
- Outside the conductor, the electric field is perpendicular to the conductor's surface.
- It has a zero charge density, ensuring that the positive and negative charges cancel each other and free charges exist only on the surface.
In addition, conductors have low resistance and high thermal conductivity. Further, a conductor placed in a magnetic field does not store energy. Finally, both ends of the conductor are at the same potential. Electricity flows through the conductor when the potential is changed at one end, which allows electrons to start flowing from one end to another.
How conductors work
According to band theory in solid-state physics, solids have a valence band and a conduction band. For a material to conduct electrical current through it, there must be no energy gap between its valence band and conduction band. Thus, in conductors, these bands overlap, allowing electrons to flow through the material even when a minimal amount of voltage is applied. Since the outer electrons in the valence band are only loosely attached to the atom, the application of voltage, an electromotive force or a thermal effect excites them, which moves them from the valence band to the conduction band.
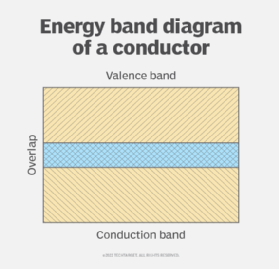
In the conduction band, these electrons can move freely anywhere, resulting in an abundance of electrons in this band. These electrons travel with a to-and-fro motion, rather than in a straight line. That's why their velocity is known as drift velocity, or Vd. It is because of this drift velocity that electrons collide with atoms of the material or other electrons inside the conductor's conduction band.
When there is a potential difference in the conductor across two points, electrons flow from the point of lower potency to the point of higher potency. Electrons and electricity flow in opposite directions. In this situation, only a small resistance is offered by the conductor material.
What are insulators?
Materials that don't allow electrical current or heat to pass through them are known as insulators, or dielectric materials. Most insulators are solid in nature. Examples include the following:
- wood
- fabric
- glass
- quartz
- mica
- plastic
- porcelain
- rubber
Most gases and some types of distilled water are also good insulators.
Resistors, semiconductors and superconductors
A material that conducts electricity fairly well but not as well as a conductor is known as a resistor. The most common example of a resistor is a combination of carbon and clay, mixed in a specific ratio to produce a constant, predictable opposition to electric current.
Semiconductors act as good conductors under some conditions but as poor conductors under others. In a semiconductor, both electrons and so-called holes -- electron absences -- act as charge carriers. Examples of semiconductors include silicon, germanium and various metal oxides.

At extremely low temperatures, some metals conduct electricity better than any known substance at room temperature. This phenomenon is called superconductivity. A substance that behaves in this way is called a superconductor.
Temperature's effects on conductivity
Temperature and conductivity are inversely related, meaning rising temperatures have an adverse effect on conduction. As temperatures increase, the vibration in conductor molecules also increases. This hampers the smooth flow of electrons, thereby decreasing the material's conductivity.
Further, rising temperatures lead to the breaking of bonds in the conductor molecules, thereby releasing electrons. This leaves the material with fewer electrons, thereby reducing the material's ability to conduct an electrical current through it.
Types of conductors
Based on their ohmic response, electrical conductors are classified as either of the following:
- ohmic conductors
- nonohmic conductors
Ohmic conductors always follow Ohm's law in which the voltage applied is directly proportional to the current flowing. Examples include aluminum, copper and silver. Nonohmic conductors, which don't follow Ohm's law, include thermistors and light-dependent resistors, or photoresistors.
Electrical conductor applications
Conductors are useful for many applications, including the following:
- Aluminum, a good conductor of heat and electricity, is commonly used to manufacture cooking utensils. It is also used in foils for food storage and preservation.
- Iron, a good conductor of heat, is used in manufacturing vehicle engines.
- Conductors are also used in automobile radiators to drive heat away from the engine.
Insulators are also used for many common applications. For instance, rubber is used to manufacture fire-resistant clothing and footwear. Plastic is frequently included in electrical appliances to prevent users from getting electrical shocks. Insulators also protect users against fire and sound.
See also: Seebeck effect, alternating current, direct current, conductance, flux, ampere, Hall effect, electromagnetic induction and impedance.
